This page provides digital assets for Nuclear Medicine Europe Members.
The Nuclear Medicine Europe’s visual assets, are tailored for the association’s Members and trusted Partners. We emphasize the importance of using these assets with utmost caution and respect. These assets are made available to highlight the strong collaboration between our association and our Members and Partners, they serve as tools for communication across various channels : website, newsletters, at industry fairs, or on social media platforms, etc.
We encourage you to utilize these assets, ensuring they accurately represent the spirit of our collaboration and the values we uphold. Together, let’s continue to elevate the field of nuclear medicine and make a meaningful impact on healthcare worldwide.
Please always use “Nuclear Medicine Europe” in full and NOT “NMEU” in your written communication.
Direct access to some of the key documents Nuclear Medicine Europe has published and where to find them.
This brief introduction provides an overview of Nuclear Medicine Europe, commonly used to introduce the association to industry partners and legislative bodies :
Nuclear Medicine Europe represents many of the major pharmaceutical & imaging equipment companies in the field of Nuclear Medicine in Europe. The combination of radiopharmaceuticals & state-of-the-art imaging permits noninvasive visualization of organs, function & structure within the body. Nuclear medicine is used in the diagnosis, management, treatment & prognosis of diseases across a broad range of medical specialities, such as oncology, cardiovascular & neurology to name a few, and as such is an integral part of patient care.
When using the logo of Nuclear Medicine Europe, it’s imperative to exercise caution and adhere to usage guidelines. This privilege is typically reserved for members of the association and partners with whom the association maintains regular collaboration. However, it’s important to note that usage by third parties outside of these categories should be approached with caution and typically requires explicit permission to ensure alignment with the association’s values and objectives.
For printable assets (vector formats), please contact David directly.
Files are in PNG format with transparency, 1600px width. click to download.
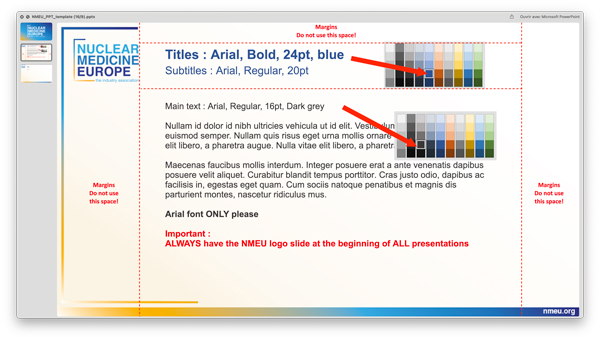
This Nuclear Medicine Europe PowerPoint template (16/9) is essentially to be used by Working Group representatives, the Executive Committee and the employees of Nuclear Medicine Europe. It consists of a series of guidelines that we ask you to follow carefully.
Within Nuclear Medicine Europe, every working group has its own distinct visual identity. This unique branding is for PowerPoint presentations, meeting minutes, reports, and any other communication materials directly associated with the workings of the respective group. By maintaining consistency and coherence across all communication channels, each working group can effectively convey its objectives, achievements, and contributions within the association.
Files are in PNG format with transparency, 1600px width. Right-click to download





whatisnuclearmedicine.com provides an illustrated introduction to nuclear medicine, aiming to educate the general public about the role of nuclear medicine in both diagnostics and therapeutics.
The site contains detailed information on how nuclear medicine works, its applications in diagnosing and treating diseases, and the significance of radiopharmaceuticals in this medical discipline.
Additionally, it provides resources such as animations, posters and guides in multiple languages, for the patients, and for free.
This platform is dedicated to providing accessible and informative content on nuclear medicine for a general audience.

The content of this page has been crafted by the Nuclear Medicine Europe Communications Working Group