2020 Activity Report
Dear members of the NMEU General Assembly,
Dear friends and colleagues,
It is hard to talk about the year 2020 without dwelling too much on the coronavirus. We all know how last year was dominated by a once-in-a-century pandemic. Nonetheless, we kept our focus on our work and on offering patients access to nuclear medicine procedures. So, our story is about what we did to support the nuclear medicine sector.
Like almost everyone in the world, we were plunged into the unknown last March. We had to improvise and to innovate. But this also allowed us to discover new resources, find new strengths and come up with new ideas.
This newsletter will not be like those of previous years. It will not show you meetings and social events that we would otherwise organise all over Europe and the United States.
But we will tell you about what else we have done. We put our energy into subjects that matter for our industry, mainly in keeping our sector going, improving our relations with the European institutions, and developing the Europe’s Beating Cancer Action Plan.
Some key European projects have opened their doors to us, notably in the field of production safety of Mo99, in regulatory affairs, and in shipping radiopharmaceuticals. Thanks to your trust and support, we completed all these successfully.
We do not know exactly what the coming months have in store for (all of) us. But we remain resolute and determined to continue doing whatever it takes to take our industry forward.
Thank you,
The Executive Committee
“Dear friends and colleagues,
We are still living under extraordinary pandemic conditions, but I hope nonetheless that you are facing the challenge with courage and optimism.
Why are we meeting today? Because our legal obligations as an association require us to maintain the mandatory points on the agenda. These include the 2019 annual accounts and the 2020 budget, as well as the validation of our newcomers.“
Read the full statement from NMEU’s President Antonis Kalemis
How nuclear medicine patient care continues in the COVID era
These are extraordinary times. Life as we knew it has been disrupted and we all have had to adapt to a new reality. Regardless, patients undergoing or awaiting life-changing medical treatments need to receive their critical care. The nuclear medicine sector is doing everything it can to make sure that people who most need interventions – diagnostic and therapeutic – can get them, despite the ongoing coronavirus pandemic.
Download “Staying safe, saving lives – How nuclear medicine patient care continues in the COVID era“
COMECER S.p.A., which has its headquarters near Bologna, Italy, has been in business since 1970. Comecer designs and manufactures high-tech systems in aseptic processing and containment for nuclear medicine. The company specialises in isolation technology solutions for regenerative medicine and tissue engineering. Comecer products are used in hospitals, universities, research centres, pharmaceutical companies and large industrial groups all over the world.
Comecer Group is represented at the NMEU General Assembly by its COO Dr. Emiliano Spagnolo.
https://www.comecer.com/
ROTEM Industries Ltd, Medical Imaging, based in Leipzig, Germany, is active in the production of consumables for radiotracers under development. Rotem has many interdisciplinary experts and enjoys close collaboration with radiopharmacies worldwide.
Rotem is represented at the NMEU General Assembly by General Manager Dr Julia Möbius.
https://www.rotemi.co.il/
ECKERT & ZIEGLER Radiopharma GmbH, based in Berlin, Germany produces pharmaceutical-grade radioisotopes, among them Ytriga, a precursor for Y-90 based oncology products; GallialPharm, a Ga-68 radionuclide generator for diagnostic applications; and other related products.
Eckert & Ziegler is represented at the NMEU General Assembly by Executive Board Member Dr Lutz Helmke.
https://radiopharma.com/
ORANO MED, based in Chatillon, France, is a nuclear and biotech company developing novel Targeted Alpha Therapies (TAT) against various types of cancers using the properties of lead-212 (212Pb) as an in-vivo generator of an alpha-emitting radioisotope. Orano Med, a subsidiary of Orano Group, relies on a robust supply chain of 212Pb, in-house expertise in chelation technologies and a preclinical facility for the development of targeted therapies. Its innovation, scientific and clinical teams believe the TAT approach has the potential to bring significant benefits where conventional therapies have failed.
Oranomed is represented at the NMEU General Assembly by President and CEO Dr Julien Dodet.
http://www.oranomed.com/
ISOVITAL – Radiopharma Logistics Group (RGL) with a registered office near Lille, France, became a full member when it acquired our Belgium member ISI. RGL specialises in the transport of radiopharmaceuticals. In recent years, their expertise has helped them become a reference in the shipment of radiopharmaceutical and sensitive materials.
RLG is represented at the NMEU General Assembly by Dr Philippe Sueur, CEO
www.radiopharmalogistics.com
Radioisotope Life Science (RLS) Inc.with a registered office in Miami, US, joined NMEU as Associate Partner on January 2, 2021. RLS recently acquired the assets of Medi-Physics, Inc. GE Healthcare’s 31 US nuclear pharmacy locations. All RLS pharmacies follow rigorous quality and safety standards with well‐established regulatory, EHS, and QA programmes, following Joint Commission accreditation for quality and safety (USP and ISO 14644‐1 standard), all of which exceed current US regulatory standards. RLS plans to expand its SPECT Radiopharmacy network, PET enabling existing sites, and PET access, while providing cutting edge radiopharmaceuticals and new molecular imaging opportunities to nuclear medicine facilities across the network.
RLS is represented at the NMEU by Chief Operating Officer Dr Shane Cobb.
https://rls.bio/
SHINE Medical Technologies was already an Associate Partner in the NMEU reactor group through their main office in the US but moved in 2019 to establish a direct presence in Europe through a Netherlands-based isotopes production facility. Now that they officially have a base in Europe, they have joined NMEU as a full member.
Shine Medical Technologies is represented at the NMEU General Assembly by Dr Harrie Buurlage, Vice President European Operations.
https://shinemed.com/
As the Covid-19 pandemic disrupted trade and transport across Europe, the European Observatory on the Supply of Medical Radioisotopes worked closely with the NMEU’s Security of Supply Working Group and the Transport Working Group to ensure the uninterrupted supply of Mo-99/Tc-99m. The Observatory also liaised with the two Working Groups on Brexit preparedness and contingency actions.
NMEU’s Emergency Response Team was instrumental in dealing with supply issues and sending information to the Observatory’s members and external stakeholders, including the EU’s Council Working Party on Atomic Questions (WPAQ), the EU Health Security Committee (HSC), the OECD Nuclear Energy Agency (NEA) and the IAEA.
In the context of Observatory work, the Euratom Supply Agency (ESA) cooperated closely on the Covid-19 response actions with the European Commission services (DG ENER, SANTE, JRC and RTD). Moreover, at a meeting of the Commission’s DG HOME Covid-19/Corona Information group, ESA debriefed the EU member states on the NMEU stakeholder concerns over the impact of the pandemic lockdowns on the transport of radioisotopes, new border controls and the cancellations of flights.

When it came to Brexit, the Observatory – through ESA – liaised with many stakeholders. Internally, these included various European Commission services (DG TRADE, TAXUD and SANTE), while externally they included the European Association of Nuclear Medicine (EANM), the British Nuclear Medicine Society (BNMS) and the Irish Nuclear Medicine Association (INMA) to raise awareness and discuss the contingency actions.
The Observatory’s spring 2020 meeting – set to be chaired jointly by NMEU and ESA – was cancelled due to Covid-19. While the autumn meeting took place during the second wave of the pandemic, it was switched from a face-to-face encounter to a virtual meeting – the first time since the Observatory was set up in 2012. As well as standard points of discussion and updates from the NMEU, OECD/NEA and EANM, the Europe’s Beating Cancer Plan was presented, along with the Commission’s SAMIRA initiative. Part of the meeting was dedicated to preparing for Brexit: the potential negative effects, mitigation arrangements as well as state of play in the UK and the Republic of Ireland.
 Last year showed, more than ever, the importance of building a strong relationship with the European Union institutions. Last February, European Commission President Ursula von der Leyen initiated a process that should lead to a new, four-year Europe’s Beating Cancer Action Plan (see below). NMEU took part in the Brussels launch of the initiative and was asked to take part in related events organized by the EU.
Last year showed, more than ever, the importance of building a strong relationship with the European Union institutions. Last February, European Commission President Ursula von der Leyen initiated a process that should lead to a new, four-year Europe’s Beating Cancer Action Plan (see below). NMEU took part in the Brussels launch of the initiative and was asked to take part in related events organized by the EU.
Nuclear medicine is already working to beat cancer by diagnosing, following up and treating it in various forms. For NMEU, liaising with the EU helps ensure the nuclear medicine sector is at the forefront of EU actions. Our links with the EU institutions also ensures we can offer valuable input about the achievements of our industries in the field. Meeting with the EU representatives and exchanging on the Europe’s Beating Cancer Action Plan shows our mutual interest and concerns for the best patient treatment.
NMEU is grateful for the excellent relationship with MEPs and the European Commission, and their ongoing interest in molecular imaging and radionuclide therapy. We are happy to take part in many EU initiatives related to the sector and to present the latest developments in the field. And we are grateful that our industry is now recognized by the EU institutions as an innovative, life-saving healthcare sector.
 NMEU was at the European Parliament on February 4, 2020, World Cancer Day, when the European Commission introduced the Europe’s Beating Cancer Action Plan.
NMEU was at the European Parliament on February 4, 2020, World Cancer Day, when the European Commission introduced the Europe’s Beating Cancer Action Plan.
Commission President Ursula von der Leyen has underlined that cancer is one of her main health priorities and that improving diagnosis and treatment will be a priority during her five-year term. Her political guidelines refer to “a European plan to fight cancer, to support Member States in improving cancer control and care, to reduce the suffering caused by this disease and for Europe to take the lead in the fight against cancer”.
The Europe’s Beating Cancer Action Plan will focus on four pillars: prevention; early diagnosis; treatment; and follow-up care. Cancer is also a key EU research and innovation priority and part of the Horizon Europe framework that began this year. Nuclear technologies will play an important role in both these initiatives.
In July 2020, NMEU was included as an expert in the European Parliament’s Beating Cancer (BECA) Committee, set up to advise the Commission during the Europe’s Beating Cancer programme. NMEU President, Antonis Kalemis gave a presentation to the BECA Committee on nuclear medicine’s innovations and the ongoing activities of our industry in Europe.
We had the opportunity to appoint three experts to the BECA Committee. These experts will advise the BECA Committee on the actions of the nuclear medicine sector in the fight against cancer.
An industry association like ours rarely receives such influence within the European institutions and we see this as an important opportunity to be able to make our voice heard with policy-makers.
When it became clear last March that the Covid-19 pandemic would disrupt the international production and supply of Mo-99 and other radioisotopes, NMEU immediately activated the Emergency Response Team (ERT). We recognised that we had to respond to emerging issues around radioisotope production and shipments, and take action to maintain secure and reliable supplies.

The ERT, which gathered members from the Security of Supply Working Group and the NMEU Transportation Working Group, held weekly teleconferences in the first months of the pandemic. Its main focus was on resolving logistical and transportation issues: many flights normally used for bulk shipments of Mo-99 and other finished radioisotopes and radiopharmaceutical products were cancelled.
With many air routes closed, the ERT worked to find new flights on passenger schedules and cargo flights, sending regular updates to the European Observatory on the Supply of Medical Radioisotopes and other international stakeholders.
The ERT also exchanged information on best practices for maintaining production and continuing to ship radioisotopes across Europe with ground transportation. The ERT finally ended its weekly teleconferences in May when cross-border travel was restored, and the logistical situation eased.
The Security of Supply Working Group (SoS WG) continued its main activities despite the challenges created by the Covid-19 pandemic. It held two meetings via videoconference, in June and November, which were shorter and more concentrated than the usual in-person meetings. But they involved increased participation, with representatives from Argentina, Japan and the European Parliament who would otherwise find it hard to take part.
The two SoS WG meetings focused on setting up coordinated reactor schedules to ensure neutron availability for both Mo-99 production and for fission co-produced Xe-133 and I-131. The coordination work for the Reactor Schedule Facilitator was ably led by Executive Committee member Erich Kollegger and was supported by key executives from the research reactors. Their work led to adjustments in the 2020 reactor schedule calendar and a re-arranged 2021 calendar that now ensures full reactor coverage throughout the year.
As well as reactor schedule coordination, the SoS WG received updates on new isotope production projects at places like Argentina (CNEA), Japan (Fuji) and the US (SHINE). The Working Group also assessed global Lu-177 production requirements; was updated on various EU initiatives including a Commission study on the development and supply of radioisotopes in the EU; examined European Parliament activities related to the sector; and discussed NMEU involvement in the Europe’s Beating Cancer Action Plan.
It is important to recognize how the SoS WG’s members and associated partners responded as the pandemic swept across Europe last March, working tirelessly to ensure there were no interruptions to production. The SoS WG includes isotope target manufacturers; research reactors; Mo-99, I-131 and Lu-177 manufacturers; Tc-99m generator manufacturers; nuclear transportation companies; and nuclear pharmacies. We have continued to successfully guarantee the secure and adequate supply of Mo-99 and other isotopes despite the second wave of the pandemic and the occasional unexpected radioisotope production facility outages.
 Nuclear Medicine Europe is now publishing frequently on Linkedin updates, articles, and news about the Nuclear Medicine industry.
Nuclear Medicine Europe is now publishing frequently on Linkedin updates, articles, and news about the Nuclear Medicine industry.
Connect to NMEU’s Linkedin account
Visitor demographics – 05 > 11 / 2020
How did the pandemic impact your day-to-day work? How do you see the next six months in the transport sector? Has this crisis made you reassess your relationship with transport companies?
NMEU’s Transport Expert Group surveyed its members to find out how it has affected the transport of radioactive material, from shipment and safety to controls and audits.
Download “How the COVID crisis has affected the transport of radiopharmaceuticals“
An initiative of the NMEU Transport Expert Working Group

NMEU was asked to join the Steering Group of a potentially crucial study on co-ordinating the development and supply of radioisotopes in the EU. This is part of the Strategic Agenda for Medical Industrial and Research Applications (SAMIRA), a European Commission initiative launched in 2017 looking at the non-power applications of nuclear and radiation technology.
The new study is named ‘Co-ordinated approach to the development and supply of radioisotopes in the EU’ (N°ENER/D3/2019-231) and will be conducted by the French company NucAdvisors.
It comes after the Commission’s Study on Sustainable and Resilient Supply of Medical Radioisotopes in the EU (SMER), completed in early 2019, which explored demand-side issues and the impact of health reimbursement mechanisms on the supply of Mo-99/Tc-99m. A SMER 2 study on PET supply has been launched and is currently in progress.
The new study aims, by the end of 2021, to define a European policy on the use of medical radioisotopes for therapy and diagnostics, which are expected to grow in significance in the coming decades. Its tasks are to:
• identify the main radioisotopes currently in use in the EU, and the main radioisotopes expected to be in use by 2030;
• identify the existing and emerging methods and technologies to produce the radioisotopes and fully describe the main elements of their respective supply chains;
• identify the main suppliers of source materials and technologies for radioisotope production and the facilities used for supply chains;
• develop scenarios and concrete options for the sustainable and secure supply of radioisotopes covered in the EU.
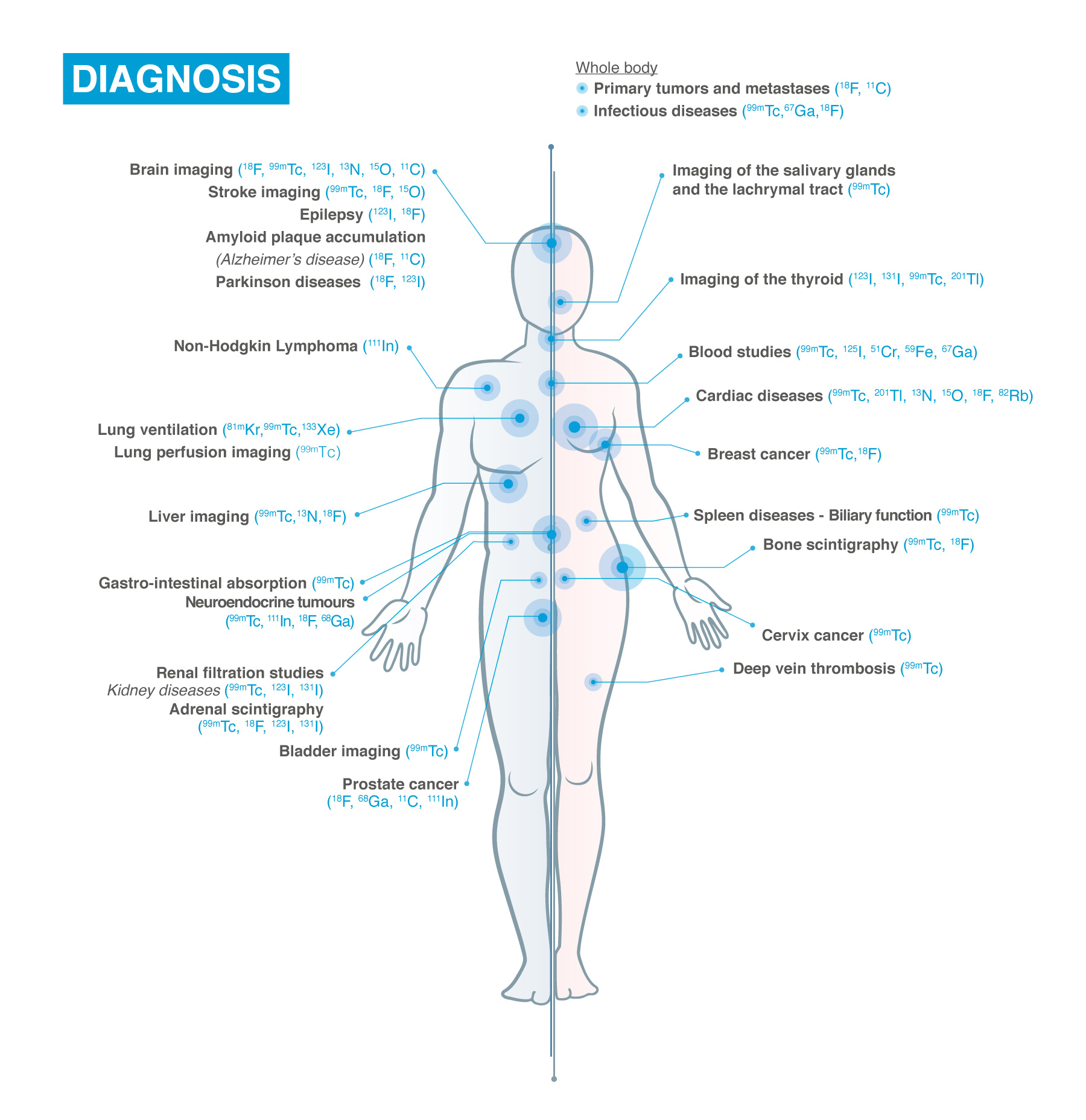
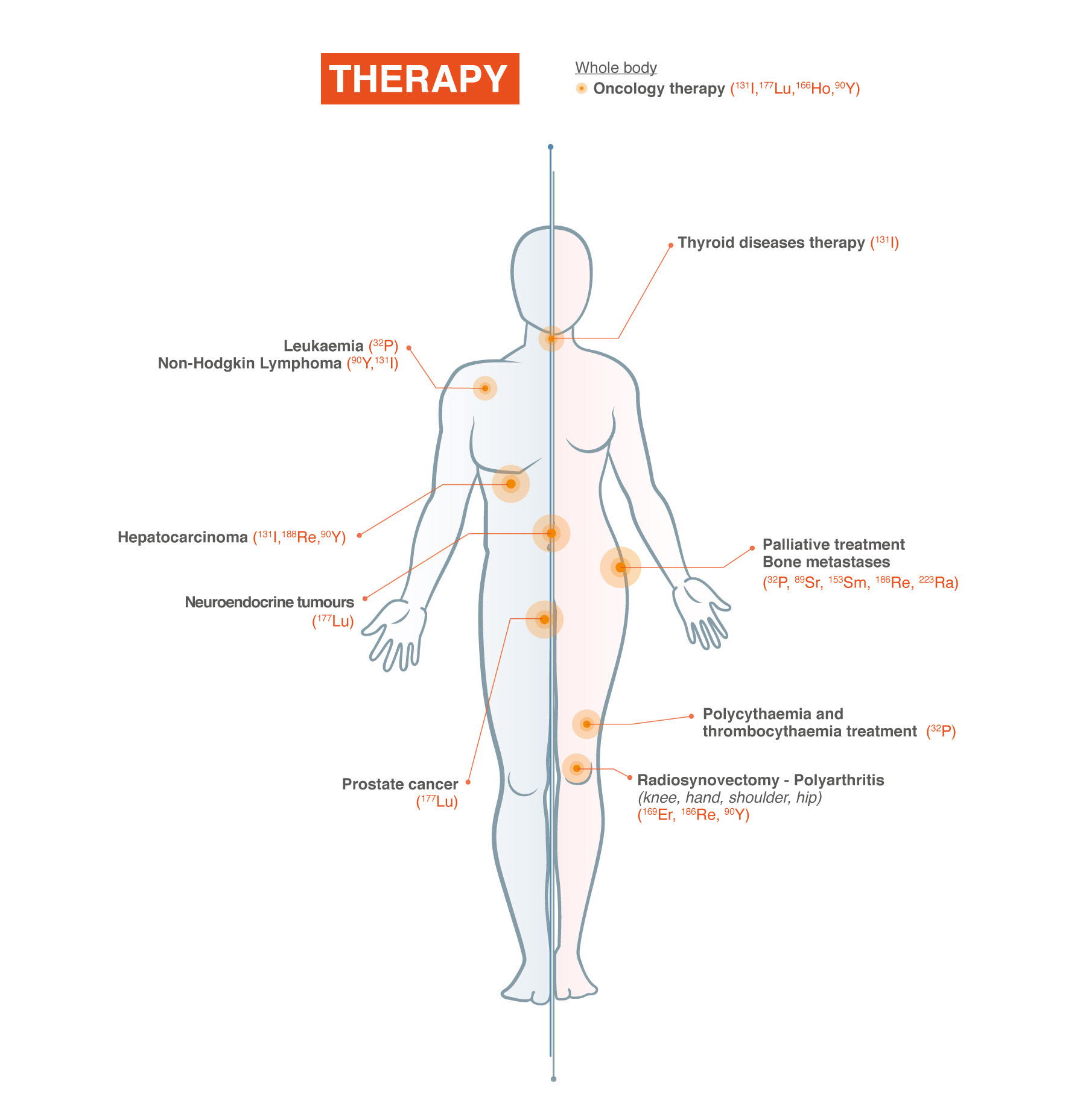
“Nuclear Medicine is based on the use of radioactivity for healthcare applications in diagnosis and therapy. Through the use of biomolecules that are labelled with radioisotopes, numerous metabolic, molecular and signalling pathways can be visualized and quantified. This information is used for diagnosis and personalized treatment planning.”
“From Status Quo to Status Go”
is now available for download.
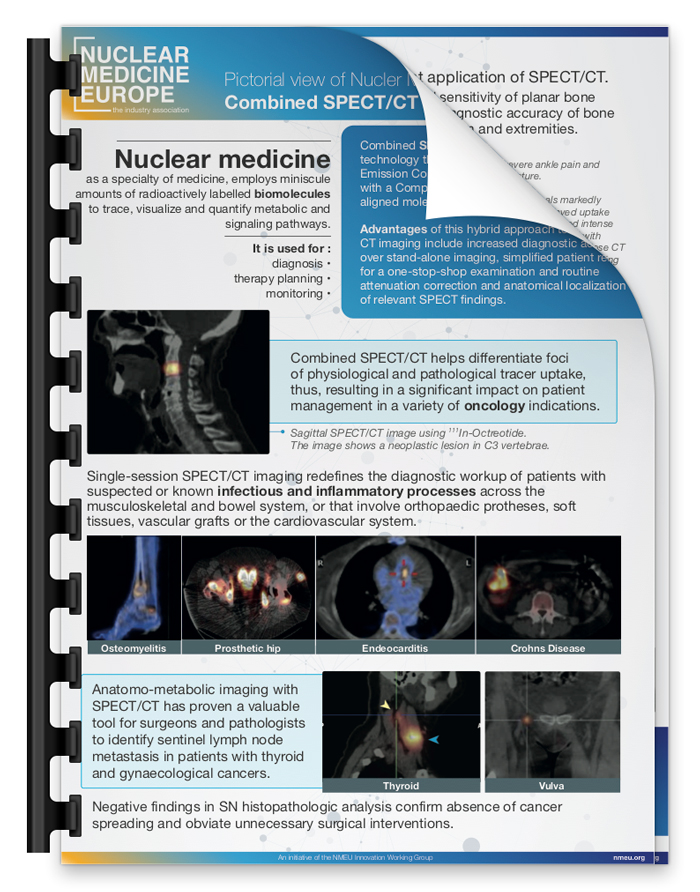
The Innovation Working Group has updated and making publicly available their Pictorial Views about Nuclear Medicine.
“Cyclotrons for Medical Radionuclide Production” and “Combined SPECT.CT imaging in clinical use” are available for download.
An initiative of the NMEU Innovation Working Group
NMEU is working with FORATOM, the European nuclear energy trade association, on a joint position paper entitled ‘Medical Uses of Nuclear Technology – Role, Challenges & Perspectives’. It will outline how nuclear technology is being used in medicine, and its current situation at EU level and in member states look like.
The paper aims to show the main challenges facing Europe’s nuclear medicine sector, the potential risks, and the opportunities. The paper will offer some detailed policy recommendations on what could be done at the EU level to ensure that a stable supply of nuclear medicine can be guaranteed and maintained within the EU.

The Executive Committee requested that Members active in targeted radionuclide therapy and theranostics should form a specific NMEU group. The group will have an advisory role in proposing activities NMEU could consider within this fast-developing field of our industry. Eleven companies expressed interest taking part in the planned Therapy Group and five of them formed a steering committee to identify areas and topics where it makes sense for NMEU to act.
Two meetings of the steering committee took place in 2020, with members agreeing to meet frequently so they can activate the group and start offering relevant advice to the Executive Committee (ExCo).
In January 2020, we announced that NMEU had joined the EMA Industry Stakeholders platform.
We then suggested making a presentation to EMA experts on radiopharmaceutical preparation and usage in Europe, which could then be used in discussions on improving regulations to help patients.
Targeted radiopharmaceuticals are already showing particular promise for the diagnosis and treatment of certain types of cancers. But despite their medical potential, there are obstacles in the EU regulatory environment to the development and take-up of targeted radiopharmaceuticals.
We have now agreed with the EMA Industry Stakeholders that a workshop on targeted radiopharmaceuticals will take place in 2021. We are extremely happy with this, which paves the way to better collaboration with EMA and new prospects for the radiopharmaceuticals industry in Europe.
Nuclear medicine and

An initiative of the NMEU Awareness Working Group
While our working groups were unable to meet face-to-face, they nevertheless communicated and worked on subjects of common interest with online tools (Teams, Skype, etc.).
The Innovation Group is now headed by Maria Martinez (Philips) and Mart Jan Blauwhoff (Curium). This group is currently working on a paper on artificial intelligence (AI) in nuclear medicine, which will soon be online.
We are happy to welcome Helen Barker (Blue Earth) as chair of the RA&Q group, accompanied by Cristiana Gameiro (IBA).
With the help of EMA, we were able to bring our members together for a webinar to jointly consider the new Brexit regulations and its consequences for our industry.
 An article in Aunt Minnie Europe, written by Leo Cendrowicz, explained in depth how the industry coped last year as the pandemic spread. In the coming days, a similar article will appear in the Politico newspaper, giving significant visibility to NMEU.
An article in Aunt Minnie Europe, written by Leo Cendrowicz, explained in depth how the industry coped last year as the pandemic spread. In the coming days, a similar article will appear in the Politico newspaper, giving significant visibility to NMEU.