Innovation


Opportunities and Challenges
“Nuclear Medicine (NM) and Artificial Intelligence (AI) together empower medical doctors for more effective patient management and better outcome. Nuclear medicine provides non-invasive, quantitative information on physiology and signalling that is essential to interpret for understanding diseases and assigning appropriate therapies.”
A computer can be programmed so that it will learn to play a better game of checkers than can be played by the person who wrote the program.
Arthur Samuel (1959)
Risks and Caveats
“AI holds a great potential to support a quantum leap of medical diagnosis and patient management. Numerous pre-clinical studies attest to the benefit of making use of computer-based processing of NM imaging. Despite these promises, a number of caveats need to be recognized”
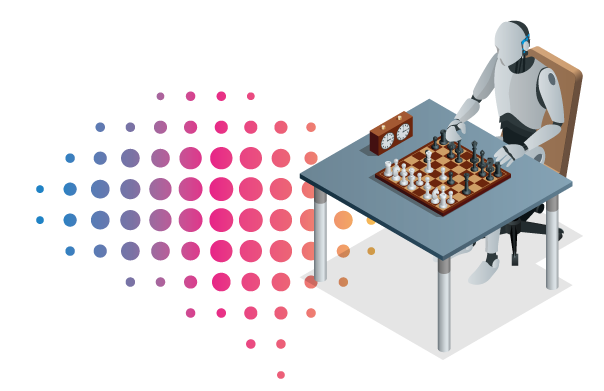

Combined SPECT/CT Imaging in Clinical use
“Nuclear medicine as a specialty of medicine employs minuscule amounts of radioactively labeled biomolecules to trace, visualize and quantify metabolic and signaling pathways.
It is used for diagnosis, therapy planning, monitoring.
Combined SPECT/CT refers to a nuclear medicine technology that physically combines a Single Photon Emission Computed Tomography (SPECT) system with a Computer Tomograph (CT) for intrinsically aligned molecular and anatomical imaging.”
From status quo to status go
“Nuclear Medicine is based on the use of radioactivity for healthcare applications in diagnosis and therapy. Through the use of biomolecules that are labelled with radioisotopes, numerous metabolic, molecular and signalling pathways can be visualized and quantified. This information is used for diagnosis and personalized treatment planning.”
Cyclotrons for Medical Radionuclide Production
“Due to the short half-life of the radionuclides required by nuclear medicine (e.g. 109 min for 18F), manufacturing centers for radiopharmaceuticals are usually associated with the cyclotron facility and all the processes including target chemistry, purification, labelling, dispensing and quality analysis is performed at the same site. This whole manufacturing process has to be repeated every day and in some cases (very short half-life) several times a day.”
Nuclear Medicine is a unique specialty of clinical medicine in modern healthcare systems. It aims at providing both diagnostic tools and therapeutic options using slightly radioactive drugs for a wide range of pathologies.
Medical need covering is supported by a broad-spectrum of technologies and innovations based on radiopharmaceuticals (radiolabelled pharmaceuticals products) and medical devices (imaging and software equipment, implantable therapeutic products).
Over the past decade a series of technological innovations in nuclear medicine allowed a predictive and more precise and more personalized patient management. Recent innovations fostered an impressive growth of “theranostic” approaches combining both diagnostic and therapeutic action within the same technology, “Innovation” is a Topical Working Group within the Nuclear Medicine Europe organization with the aim to provide a gateway to detecting and understanding innovation in the field of non-invasive imaging and therapies involving broad-spectrum nuclear medicine technologies. Selected experts of member companies from imaging and pharmaceutical industries set up the Innovation Working Group joining their extensive experience in applied and advanced imaging technology.
The Group receives input from its members on current innovations and associated developments worldwide., Together with the NMEU Executive Committee, WG Innovation highlights promising developments to all Nuclear Medicine Europe members.
In view of the rapid advances and significant changes in nuclear medicine, the group develops educational activities and provides teaching materials for the Nuclear Medicine Europe related-communities: patients, medical/nuclear physicians and decision makers/influencers. In close collaboration with the NMEU Executive Committee, the group monitors EU Research initiatives and advises NMEU Members on potential support activities of these initiatives.
Furthermore, the group informs the NMEU members on topics of interest from the nuclear medicine community and initiates as well as supports survey activities.
WG Innovation liaises closely with complementary working groups or committees within other European organizations to actively support nuclear medicine innovation, training and development with a particular focus on projects and strategies towards personalized, medicine.
Maria-Jose Martinez, Chair, Philips, The Netherlands
Mart Jan Blauwhoff, Vice Chairman Curium The Netherlands
Jean Michel Geets, IBA Radiopharma Solutions, Belgium
Florence Chossat Curium France
Jean-François Gestin Atlanpole Biotherapies, France
Fabrice Giacomelli Nucleis, Belgium
Ira Goldman Lantheus Medical Imaging, Ireland
Angel Hijos Lilly, France
Jean-Luc Morelle Trasis, Belgium
Stanley Satz AIP- Advance Innovative Partners, Germany
Ayfer A. Akit Monrol, Romania
Ewout VanSteenkiste Molecubes, Belgium
Samuel Voccia ANMI Telix, Belgium
Nissan Leepaz Product Development Manager, Medical Imaging – Rotem Europe
Matteo Melandri, Comecer Radiopharma Division – Comecer, Italy
Hartwig Newiger, Siemens Healthineers
Antonis Kalemis, President NMEu & Group Supervisor Siemens Healthineers
Ilyes Zahi, AAA France